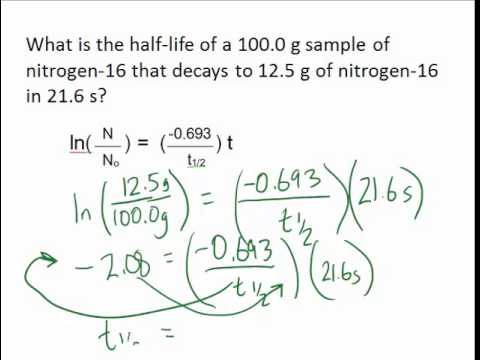
For a first-order reaction, the half life of the reactant may be called the half life of the reaction. For a reactant that is entirely consumed it is the time taken for the reactant concentration to fall to one half of its initial value. 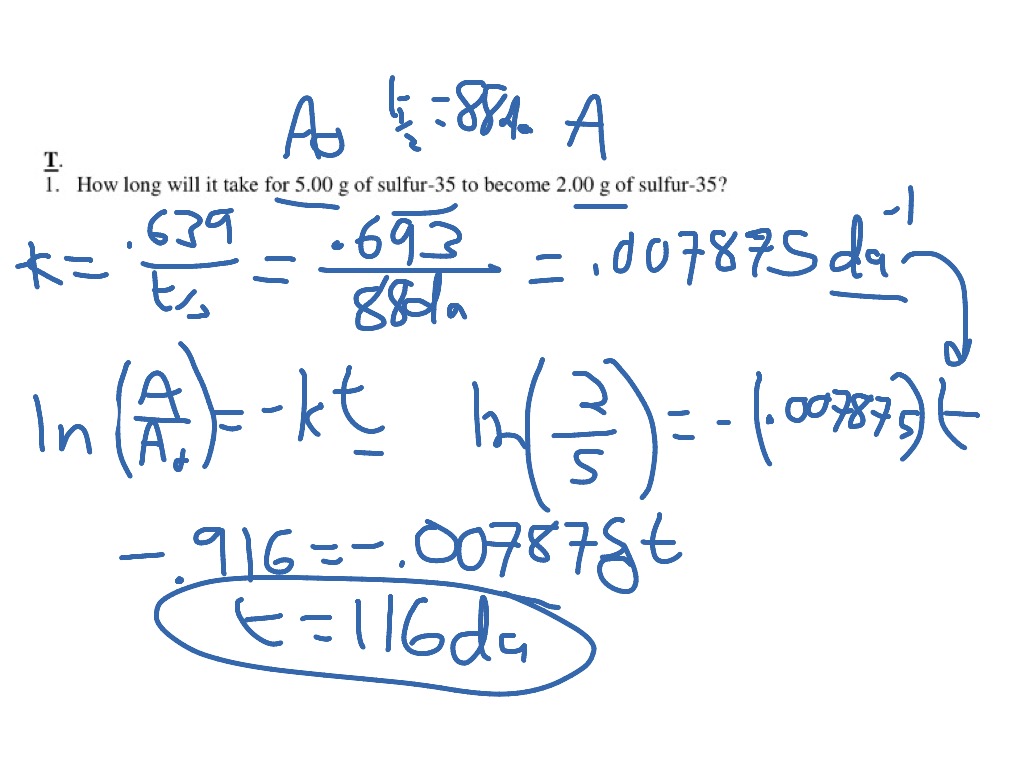
Definition for Half-Life :The time it takes for one-half of the atoms of an unstable element or nuclide to decay radioactively into another element or nuclideįor a given reaction the half life t1/2 of a reactant is the time required for its concentration to reach a value that is the arithmetic mean of its initial and final (equilibrium) value. The time required for half of a given sample to undergo radioactive decay. 
The time required for half of a reactant to be converted into product(s). The amount of time it takes for half an initial amount to disintegrate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
